Transgender hormone therapy
 From Wikipedia the free encyclopedia
From Wikipedia the free encyclopedia
| Part of a series on |
| Transgender topics |
|---|
| |
Transgender hormone therapy, also called hormone replacement therapy (HRT) or gender-affirming hormone therapy (GAHT), is a form of hormone therapy in which sex hormones and other hormonal medications are administered to transgender or gender nonconforming individuals for the purpose of more closely aligning their secondary sexual characteristics with their gender identity. This form of hormone therapy is given as one of two types, based on whether the goal of treatment is masculinization or feminization:
- Masculinizing hormone therapy – for transgender men or transmasculine people; consists of androgens and antiestrogens.
- Feminizing hormone therapy – for transgender women or transfeminine people; consists of estrogens with or without antiandrogens.
Eligibility for transgender hormone therapy may be concluded by assessing a patient for gender dysphoria or persistent gender incongruence, though many medical institutions now used an informed consent model. This model ensures patients are informed of the procedure process, including possible benefits and risks, while removing many of the historical barriers needed to start hormone therapy. Treatment guidelines for therapy have been developed by several medical associations.
Some intersex people may also undergo hormone therapy, either starting in childhood to confirm the sex they were assigned at birth, or later in order to align their sex with their gender identity. Non-binary people may also engage in hormone therapy in order to achieve a desired balance of sex hormones or to help align their bodies with their gender identities.[1] Many transgender people obtain hormone therapy from a licensed health care provider and others obtain and self-administer hormones.
Requirements[edit]
The formal requirements to begin gender-affirming hormone therapy vary widely depending on geographic location and specific institution. Gender affirming hormones can be prescribed by a wide range of medical providers including, but not limited to, primary care physicians, endocrinologists, and obstetrician-gynecologists.[2]
Historically, many health centers required a psychiatric evaluation and/or a letter from a therapist before beginning therapy. Many centers now use an informed consent model that does not require any routine formal psychiatric evaluation but instead focuses on reducing barriers to care by ensuring a person can understand the risks, benefits, alternatives, unknowns, limitations, and risks of no treatment.[3] Some LGBT health organizations (notably Chicago's Howard Brown Health Center[4] and Planned Parenthood[5]) advocate for this type of informed consent model.
The World Professional Association for Transgender Health (WPATH) Standards of Care, 7th edition, note that both of these approaches to care are appropriate.[2]
Gender dysphoria[edit]
Many international guidelines and institutions require persistent, well-documented gender dysphoria as a pre-requisite to starting gender-affirmation therapy. Gender dysphoria refers to the psychological discomfort or distress that an individual can experience if their sex assigned at birth is incongruent with that person's gender identity.[6] Signs of gender dysphoria can include comorbid mental health stressors such as depression, anxiety, low self-esteem, and social isolation.[7] Not all gender nonconforming individuals experience gender dysphoria.[8]
Treatment options[edit]
Guidelines[edit]
For transgender youth, the Dutch protocol existed as among the earlier guidelines for hormone therapy by delaying puberty until age 16.[9][10] The World Professional Association for Transgender Health (WPATH) and the Endocrine Society later formulated guidelines that created a foundation for health care providers to care for transgender patients.[11][12] UCSF guidelines are also sometimes used.[3] There is no generally agreed-upon set of guidelines, however.[13]
Delaying puberty in adolescents[edit]
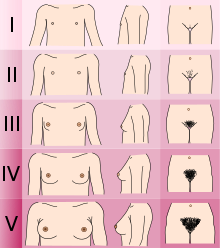
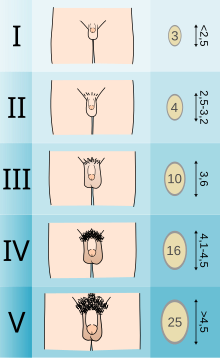
Adolescents experiencing gender dysphoria may opt to undergo puberty-suppressing hormone therapy at the onset of puberty. The Standards of Care set forth by WPATH recommend individuals pursuing puberty-suppressing hormone therapy wait until at least experiencing Tanner Stage 2 pubertal development.[6] Tanner Stage 2 is defined by the appearance of scant pubic hair, breast bud development, and/or slight testicular growth.[14] WPATH classifies puberty-suppressing hormone therapy as a "fully reversible" intervention. Delaying puberty allows individuals more time to explore their gender identity before deciding on more permanent interventions and prevents the physical changes associated with puberty.[6]
The preferred puberty-suppressing agent for both individuals assigned male at birth and individuals assigned female at birth is a GnRH Analogue.[6] This approach temporarily shuts down the Hypothalamic-Pituitary-Gonadal (HPG) Axis, which is responsible for the production of hormones (estrogen, testosterone) that cause the development of secondary sexual characteristics in puberty.[15]
Feminizing hormone therapy[edit]
Feminizing hormone therapy is typically used by transgender women, who desire the development of feminine secondary sex characteristics. Individuals who identify as non-binary may also opt-in for feminizing hormone treatment to better align their body with their desired gender expression.[16] Feminizing hormone therapy usually includes medication to suppress testosterone production and induce feminization. Types of medications include estrogens, antiandrogens (testosterone blockers), and progestogens.[17] Most commonly, an estrogen is combined with an antiandrogen to suppress and block testosterone.[18] This allows for demasculinization and promotion of feminization and breast development. Estrogens are administered in various modalities including injection, transdermal patch, and oral tablets.[18]
The desired effects of feminizing hormone therapy focus on the development of feminine secondary sex characteristics. These desired effects include: breast tissue development, redistribution of body fat, decreased body hair, reduction of muscle mass, and more.[18] The table below summarizes some of the effects of feminizing hormone therapy in transgender women:
| Effect | Time to expected onset of effect[a] | Time to expected maximum effect[a][b] | Permanency if hormone therapy is stopped |
|---|---|---|---|
| Breast development and nipple/areolar enlargement | 2–6 months | 1–5 years | Permanent |
| Thinning/slowed growth of facial/body hair | 4–12 months | >3 years[c] | Reversible |
| Cessation/reversal of male-pattern scalp hair loss | 1–3 months | 1–2 years[d] | Reversible |
| Softening of skin/decreased oiliness and acne | 3–6 months | Unknown | Reversible |
| Redistribution of body fat in a feminine pattern | 3–6 months | 2–5 years | Reversible |
| Decreased muscle mass/strength | 3–6 months | 1–2 years[e] | Reversible |
| Widening and rounding of the pelvis[f] | Unspecified | Unspecified | Permanent |
| Changes in mood, emotionality, and behavior | Unspecified | Unspecified | Reversible |
| Decreased sex drive | 1–3 months | Temporary[19] | Reversible |
| Decreased spontaneous/morning erections | 1–3 months | 3–6 months | Reversible |
| Erectile dysfunction and decreased ejaculate volume | 1–3 months | Variable | Reversible |
| Decreased sperm production/fertility | Unknown | >3 years | Reversible or permanent[g] |
| Decreased testicle size | 3–6 months | 2–3 years | Unknown |
| Decreased penis size | None[h] | Not applicable | Not applicable |
| Decreased prostate gland size | Unspecified | Unspecified | Unspecified |
| Voice changes | None[i] | Not applicable | Not applicable |
Footnotes and sources Footnotes:
| |||
Masculinizing hormone therapy[edit]
Masculinizing hormone therapy is typically used by transgender men, who desire the development of masculine secondary sex characteristics. Masculinizing hormone therapy usually includes testosterone to produce masculinization and suppress the production of estrogen.[32] Treatment options include oral, parenteral, subcutaneous implant, and transdermal (patches, gels). Dosing is patient-specific and is discussed with the physician.[33] The most commonly prescribed methods are intramuscular and subcutaneous injections. This dosing can be weekly or biweekly depending on the individual patient.[citation needed]
Unlike feminizing hormone therapy, individuals undergoing masculinizing hormone therapy do not usually require additional hormone suppression such as estrogen suppression. Therapeutic doses of testosterone are usually sufficient to inhibit the production of estrogen to desired physiologic levels.[15]
The desired effects of masculinizing hormone therapy focus on the development of masculine secondary sex characteristics. These desired effects include: increased muscle mass, development of facial hair, voice deepening, increase and thickening of body hair, and more.[34]
| Reversible Changes | Irreversible Changes |
|---|---|
| Increased libido | Deepening of voice |
| Redistribution of body fat | Growth of facial/body hair |
| Cessation of ovulation/menstruation | Male-pattern baldness |
| Increased muscle mass | Enlargement of clitoris |
| Increased perspiration | Growth spurt/closure of growth plates |
| Acne | Breast atrophy |
| Increased RBC count |
Safety[edit]
Hormone therapy for transgender individuals has been shown in medical literature to be generally safe, when supervised by a qualified medical professional.[35] There are potential risks with hormone treatment that will be monitored through screenings and lab tests such as blood count (hemoglobin), kidney and liver function, blood sugar, potassium, and cholesterol.[33][17] Taking more medication than directed may lead to health problems such as increased risk of cancer, heart attack from thickening of the blood, blood clots, and elevated cholesterol.[33][36]
Feminizing hormone therapy[edit]
The Standards of Care published by the World Professional Association for Transgender Health (WPATH) summarize many of the risks associated with feminizing hormone therapy (outlined below).[6] For more in-depth information on the safety profile of estrogen-based feminizing hormone therapy visit the feminizing hormone therapy page.
| Likely Increased Risk | Possible Increased Risk | Inconclusive/No Increased Risk |
|---|---|---|
| Venous thromboembolic disease | Type 2 diabetes | Breast cancer |
| Cardiovascular disease | Hypertension | Prostate cancer |
| Hypertriglyceridemia | Hyperprolactinaemia | |
| Gallstones | Osteoporosis | |
| Hyperkalemia | ||
| Cerebrovascular disease | ||
| Polyuria (or dehydration)[a] | ||
| Meningioma[b] | ||
| ||
Masculinizing hormone therapy[edit]
The Standards of Care published by the World Professional Association for Transgender Health (WPATH) summarize many of the risks associated with masculinizing hormone therapy (outlined below).[6] For more in-depth information on the safety profile of testosterone-based masculinizing hormone therapy visit the masculinizing hormone therapy page.
| Likely Increased Risk | Possible Increased Risk | Inconclusive/No Increased Risk |
|---|---|---|
| Polycythemia | Type 2 diabetes | Osteoporosis |
| Weight gain | Breast cancer | |
| Acne | Ovarian cancer | |
| Pattern hair loss | Uterine cancer | |
| Hypertension | Cervical cancer | |
| Sleep apnea | ||
| Decreased HDL cholesterol | ||
| Decreased LDL cholesterol | ||
| Cardiovascular disease | ||
| Hypertriglyceridemia |
Fertility consideration[edit]
Transgender hormone therapy may limit fertility potential.[38] Should a transgender individual choose to undergo sex reassignment surgery, their fertility potential is lost completely.[39] Before starting any treatment, individuals may consider fertility issues and fertility preservation. Options include semen cryopreservation, oocyte cryopreservation, and ovarian tissue cryopreservation.[38][39]
A study presented at ENDO 2019 (the Endocrine Society's conference) shows that even after one year of treatment with testosterone, a transgender man can preserve his fertility potential.[40]
Treatment eligibility[edit]
Many providers use informed consent, whereby someone seeking hormone therapy can sign a statement of informed consent and begin treatment without much gatekeeping. For other providers, eligibility is determined using major diagnostic tools such as ICD-11 or the Diagnostic and Statistical Manual of Mental Disorders (DSM) to classify a patient with gender dysphoria. Psychiatric conditions can commonly accompany or present similar to gender incongruence and gender dysphoria. For this reason, patients are assessed using DSM-5 criteria or ICD-11 criteria in addition to screening for psychiatric disorders. The Endocrine Society requires physicians that diagnose gender dysphoria and gender incongruence to be trained in psychiatric disorders with competency in ICD-11 and DSM-5. The healthcare provider should also obtain a thorough assessment of the patient's mental health and identify potential psychosocial factors that can affect therapy.[41]
WPATH Standards of Care[edit]
The WPATH Standards of Care, most recently published in 2022, outlines a series of guidelines which should be met before a patient should be allowed transgender hormone replacement therapy:[37]
- Gender incongruence is marked and sustained
- Patient meets diagnostic criteria for gender incongruence prior to gender-affirming hormone treatment in regions where a diagnosis is necessary to access health care
- Patient has capacity to consent to hormone therapy treatment
- Other possible causes of apparent gender incongruence have been identified and excluded
- Mental health and physical conditions that could negatively impact the outcome of treatment have been assessed
- Understands the effect of gender-affirming hormone treatment on reproduction and they have explored reproductive options
Readiness[edit]
Some organizations – but fewer than in the past – require that patients spend a certain period of time living in their desired gender role before starting hormone therapy. This period is sometimes called real-life experience (RLE).
In Sweden, for instance, patients seeking to access gender affirming healthcare must first undergo extended evaluations with psychiatric professionals, during which they must - without any form of medical transition - successfully live for one full year as their desired gender in all professional, social, and personal matters. Gender clinics are recommended to provide patients with wigs and breast prostheses for the endeavor. The evaluation additionally involves, if possible, meetings with family members and/or other individuals close to the patient. Patients may be denied care for any number of "psychosocial dimensions", including their choice of job or their marital status.[42][43]
Transgender and gender non-conforming activists, such as Kate Bornstein, have asserted that RLE is psychologically harmful and is a form of "gatekeeping", effectively barring individuals from transitioning for as long as possible, if not permanently.[44]
In September 2022, the World Professional Association for Transgender Health (WPATH) Standards of Care for the Health of Transgender and Gender Diverse People (SOC) Version 8 were released and removed the requirement of RLE for all gender-affirming treatments, including gender-affirming surgery.[45]
Accessibility[edit]
Gender-affirming care is health care that affirms people to live authentically in their genders, no matter the gender they were assigned at birth or the path their gender affirmation (or transition) takes. It allows each person to seek only the changes or medical interventions they desire to affirm their own gender identity, and hormone therapy ("HRT" or gender-affirming hormone therapy) may be a part of that.[46]
Some transgender people choose to self-administer hormone replacement medications, often because doctors have too little experience in this area, or because no doctor is available. Others self-administer because their doctor will not prescribe hormones without an approval letter from a psychotherapist. Many therapists require extended periods of continuous psychotherapy and/or real-life experience before they will write such a letter. Because many individuals must pay for evaluation and care out-of-pocket, costs can be prohibitive.[citation needed]
Access to medication can be poor even where health care is provided free. In a patient survey conducted by the United Kingdom's National Health Service in 2008, 5% of respondents acknowledged resorting to self-medication, and 46% were dissatisfied with the amount of time it took to receive hormone therapy. The report concluded in part: "The NHS must provide a service that is easy to access so that vulnerable patients do not feel forced to turn to DIY remedies such as buying drugs online with all the risks that entails. Patients must be able to access professional help and advice so that they can make informed decisions about their care, whether they wish to take the NHS or private route without putting their health and indeed their lives in danger."[47] Self-administration of hormone replacement medications without medical supervision may have untoward health effects and risks.[48]
A number of private companies have attempted to increase accessibility for hormone replacement medications and help transgender people navigate the complexities of access to treatment.[citation needed]
See also[edit]
References[edit]
- ^ Ferguson JM (November 30, 2017). "What It Means to Transition When You're Non-Binary". Teen Vogue.
- ^ a b Deutsch MB, Feldman JL (January 2013). "Updated recommendations from the world professional association for transgender health standards of care". American Family Physician. 87 (2): 89–93. PMID 23317072.
- ^ a b c Deutsch MB, ed. (June 2016). Guidelines for the Primary and Gender-Affirming Care of Transgender and Gender Nonbinary People (2nd ed.). San Francisco, CA: UCSF Transgender Care, Department of Family and Community Medicine, University of California San Francisco.
- ^ Schreiber L. "Howard Brown Health Center Establishes Transgender Hormone Protocol". www.howardbrown.org. Howard Brown. Archived from the original on 2011-10-08. Retrieved 2011-08-25.
- ^ "What Health Care & Services Do Transgender People Require?". www.plannedparenthood.org. Retrieved 2019-10-16.
- ^ a b c d e f g h Coleman E, Bockting W, Botzer M, Cohen-Kettenis P, DeCuypere G, Feldman J, et al. (August 2012). "Standards of Care for the Health of Transsexual, Transgender, and Gender-Nonconforming People, Version 7". International Journal of Transgenderism. 13 (4): 165–232. doi:10.1080/15532739.2011.700873. S2CID 39664779.
- ^ "Gender dysphoria". nhs.uk. 2017-10-23. Retrieved 2021-11-15.
- ^ Olson-Kennedy J, Cohen-Kettenis PT, Kreukels BP, Meyer-Bahlburg HF, Garofalo R, Meyer W, Rosenthal SM (April 2016). "Research priorities for gender nonconforming/transgender youth: gender identity development and biopsychosocial outcomes". Current Opinion in Endocrinology, Diabetes, and Obesity. 23 (2): 172–179. doi:10.1097/MED.0000000000000236. PMC 4807860. PMID 26825472.
- ^ Shumer, Daniel E.; Spack, Norman P. (January 2015). "Paediatrics: Transgender medicine--long-term outcomes from 'the Dutch model'". Nature Reviews. Urology. 12 (1): 12–13. doi:10.1038/nrurol.2014.316. ISSN 1759-4820. PMC 4349440. PMID 25403246.
- ^ Delemarre-van de Waal, Henriette A; Cohen-Kettenis, Peggy T (November 2006). "Clinical management of gender identity disorder in adolescents: a protocol on psychological and paediatric endocrinology aspects". European Journal of Endocrinology. 155 (suppl_1): S131–S137. doi:10.1530/eje.1.02231. ISSN 0804-4643.
- ^ Hembree, Wylie C.; Cohen-Kettenis, Peggy; Delemarre-van de Waal, Henriette A.; Gooren, Louis J.; Meyer, Walter J.; Spack, Norman P.; Tangpricha, Vin; Montori, Victor M. (2009-09-01). "Endocrine Treatment of Transsexual Persons:An Endocrine Society Clinical Practice Guideline". The Journal of Clinical Endocrinology & Metabolism. 94 (9): 3132–3154. doi:10.1210/jc.2009-0345. ISSN 0021-972X.
- ^ Unger CA (December 2016). "Hormone therapy for transgender patients". Translational Andrology and Urology. 5 (6): 877–884. doi:10.21037/tau.2016.09.04. PMC 5182227. PMID 28078219.
- ^ Houssayni, Sarah; Nilsen, Kari (Feb 28, 2018). "Transgender Competent Provider: Identifying Transgender Health Needs, Health Disparities, and Health Coverage". Kansas Journal of Medicine. 11 (1): 15–19. doi:10.17161/kjm.v11i1.8679. PMC 5834239. PMID 29844850.
- ^ Emmanuel M, Bokor BR (2021). "Tanner Stages". StatPearls. Treasure Island (FL): StatPearls Publishin. PMID 29262142. Retrieved 2021-11-12.
- ^ a b Bangalore Krishna K, Fuqua JS, Rogol AD, Klein KO, Popovic J, Houk CP, et al. (2019). "Use of Gonadotropin-Releasing Hormone Analogs in Children: Update by an International Consortium". Hormone Research in Paediatrics. 91 (6): 357–372. doi:10.1159/000501336. PMID 31319416. S2CID 197664792.
- ^ "Hormone Use for Non-Binary People". GenderGP. Retrieved 2020-10-18.
- ^ a b "Information on Estrogen Hormone Therapy". Transgender Care. transcare.ucsf.edu. Retrieved 2019-08-07.
- ^ a b c "Overview of feminizing hormone therapy". Gender Affirming Health Program. transcare.ucsf.edu. Retrieved 2021-11-12.
- ^ Defreyne J, Elaut E, Kreukels B, Daphne Fisher A, Castellini G, Staphorsius A, Den Heijer M, Heylens G, T'Sjoen G (April 2020). "Sexual Desire Changes in Transgender Individuals Upon Initiation of Hormone Treatment: Results From the Longitudinal European Network for the Investigation of Gender Incongruence". The Journal of Sexual Medicine. 17 (4): 812–825. doi:10.1016/j.jsxm.2019.12.020. PMID 32008926.
- ^ Elliott S, Latini DM, Walker LM, Wassersug R, Robinson JW (September 2010). "Androgen deprivation therapy for prostate cancer: recommendations to improve patient and partner quality of life". The Journal of Sexual Medicine. 7 (9): 2996–3010. doi:10.1111/j.1743-6109.2010.01902.x. PMID 20626600.
- ^ Higano CS (February 2003). "Side effects of androgen deprivation therapy: monitoring and minimizing toxicity". Urology. 61 (2 Suppl 1): 32–38. doi:10.1016/S0090-4295(02)02397-X. PMID 12667885.
- ^ Higano CS (October 2012). "Sexuality and intimacy after definitive treatment and subsequent androgen deprivation therapy for prostate cancer". Journal of Clinical Oncology. 30 (30): 3720–3725. doi:10.1200/JCO.2012.41.8509. PMID 23008326.
- ^ Nieschlag E, Behre H (29 June 2013). Andrology: Male Reproductive Health and Dysfunction. Springer Science & Business Media. pp. 54–. ISBN 978-3-662-04491-9.
- ^ Hembree WC, Cohen-Kettenis PT, Gooren L, Hannema SE, Meyer WJ, Murad MH, et al. (November 2017). "Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline". The Journal of Clinical Endocrinology and Metabolism. 102 (11): 3869–3903. doi:10.1210/jc.2017-01658. PMID 28945902. S2CID 3726467.
- ^ Bourns A (2015). "Guidelines and Protocols for Comprehensive Primary Care for Trans Clients" (PDF). Sherbourne Health Centre. Retrieved 15 August 2018.
- ^ Fabris B, Bernardi S, Trombetta C (March 2015). "Cross-sex hormone therapy for gender dysphoria". Journal of Endocrinological Investigation. 38 (3): 269–282. doi:10.1007/s40618-014-0186-2. hdl:11368/2831597. PMID 25403429. S2CID 207503049.
- ^ Moore E, Wisniewski A, Dobs A (August 2003). "Endocrine treatment of transsexual people: a review of treatment regimens, outcomes, and adverse effects". The Journal of Clinical Endocrinology and Metabolism. 88 (8): 3467–3473. doi:10.1210/jc.2002-021967. PMID 12915619.
- ^ Asscheman H, Gooren LJ (1993). "Hormone Treatment in Transsexuals". Journal of Psychology & Human Sexuality. 5 (4): 39–54. doi:10.1300/J056v05n04_03. ISSN 0890-7064. S2CID 144580633.
- ^ Levy A, Crown A, Reid R (October 2003). "Endocrine intervention for transsexuals". Clinical Endocrinology. 59 (4): 409–418. doi:10.1046/j.1365-2265.2003.01821.x. PMID 14510900. S2CID 24493388.
- ^ de Blok C, Klaver M, Nota N, Dekker M, den Heijer M (2016). "Breast development in male-to-female transgender patients after one year cross-sex hormonal treatment". Endocrine Abstracts. doi:10.1530/endoabs.41.GP146. ISSN 1479-6848.
- ^ de Blok CJ, Klaver M, Wiepjes CM, Nota NM, Heijboer AC, Fisher AD, et al. (February 2018). "Breast Development in Transwomen After 1 Year of Cross-Sex Hormone Therapy: Results of a Prospective Multicenter Study". The Journal of Clinical Endocrinology and Metabolism. 103 (2): 532–538. doi:10.1210/jc.2017-01927. PMID 29165635. S2CID 3716975.
- ^ "Masculinizing hormone therapy - Mayo Clinic". www.mayoclinic.org. Retrieved 2019-08-02.
- ^ a b c "Information on Testosterone Hormone Therapy". Transgender Care. transcare.ucsf.edu. Retrieved 2019-08-07.
- ^ Deutsch MB (17 June 2016). "Overview of masculinizing hormone therapy". UCSF Gender Affirming Health Program. San Francisco, CA: The University of California. Retrieved 2021-11-12.
- ^ Weinand JD, Safer JD (June 2015). "Hormone therapy in transgender adults is safe with provider supervision; A review of hormone therapy sequelae for transgender individuals". Journal of Clinical & Translational Endocrinology. 2 (2): 55–60. doi:10.1016/j.jcte.2015.02.003. PMC 5226129. PMID 28090436.
- ^ "A randomized, double-blind study of two combined oral contraceptives containing the same progestogen, but different estrogens. World Health Organization Task Force on Oral Contraception". Contraception. 21 (5): 445–459. May 1980. doi:10.1016/0010-7824(80)90010-4. PMID 7428356.
- ^ a b c Coleman E, Radix AE, Bouman WP, Brown GR, de Vries AL, Deutsch MB, et al. (2022-08-19). "Standards of Care for the Health of Transgender and Gender Diverse People, Version 8". International Journal of Transgender Health. 23 (Suppl 1): S1–S259. doi:10.1080/26895269.2022.2100644. PMC 9553112. PMID 36238954.
- ^ a b T'Sjoen G, Van Caenegem E, Wierckx K (December 2013). "Transgenderism and reproduction". Current Opinion in Endocrinology, Diabetes, and Obesity. 20 (6): 575–579. doi:10.1097/01.med.0000436184.42554.b7. PMID 24468761. S2CID 205398449.
- ^ a b De Sutter P (April 2001). "Gender reassignment and assisted reproduction: present and future reproductive options for transsexual people". Human Reproduction. 16 (4): 612–614. doi:10.1093/humrep/16.4.612. PMID 11278204.
- ^ "Ovary function is preserved in transgender men at one year of testosterone therapy". Endocrine Society. 23 March 2019. Retrieved 25 March 2019.
- ^ "CORRIGENDUM FOR "Endocrine Treatment of Gender-Dysphoric/Gender-Incongruent Persons: An Endocrine Society Clinical Practice Guideline"". The Journal of Clinical Endocrinology and Metabolism. 103 (7): 2758–2759. July 2018. doi:10.1210/jc.2018-01268. PMID 29905821.
- ^ Linander, Ida; Lauri, Marcus (2021). "Two Steps Forward, One Step Back: A Policy Analysis of the Swedish Guidelines for Trans-Specific Healthcare". Sexuality Research and Social Policy. 18 (2): 309–320. doi:10.1007/s13178-020-00459-5. S2CID 256073192.
- ^ "God vård av vuxna med könsdysfori" (PDF).
- ^ Bornstein, Kate (2013). My Gender Workbook, Updated : How to Become a Real Man, a Real Woman, the Real You, or Something Else Entirely (2nd ed.). New York: Routledge. ISBN 978-0415538657.
- ^ Coleman, E.; et al. (19 August 2022). "Standards of Care for the Health of Transgender and Gender Diverse People, Version 8". International Journal of Transgender Health. 23 (sup1): S1–S259. doi:10.1080/26895269.2022.2100644. ISSN 2689-5269. PMC 9553112. PMID 36238954.
- ^ "Gender-Affirming Hormone Therapy 101: Introducing the #HRTSavesLives Campaign". 10 August 2020.
- ^ "Survey of Patient Satisfaction with Transgender Services" (PDF). The Audit Information & Analysis Unit. National Health Service. Archived from the original (PDF) on 2016-03-04. Retrieved 2016-01-08.
- ^ Becerra Fernández A, de Luis Román DA, Piédrola Maroto G (October 1999). "[Morbidity in transsexual patients with cross-gender hormone self-treatment]" [Morbidity in transsexual patients with cross-gender hormone self-treatment] (PDF). Medicina Clinica (in Spanish). 113 (13): 484–487. PMID 10604171. Archived from the original (PDF) on 2017-12-08. Retrieved 2018-11-11.