Hydroacylation
 From Wikipedia the free encyclopedia
From Wikipedia the free encyclopedia
Hydroacylation is a type of organic reaction in which an electron-rich[1] unsaturated hydrocarbon inserts into a formyl C-H bond. With alkenes, the product is a ketone:
- RCHO + CH2=CHR' → RC(O)CH2CH2R'
With an alkyne instead, the reaction produces an α,β-unsaturated ketone.[2]
The reaction requires a metal catalyst or a radical initiator.[1] It is almost invariably practiced as an intramolecular reaction using homogeneous catalysts, often based on rhodium phosphines.
History[edit]
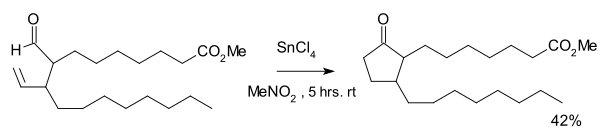
The reaction was discovered in the 1970s as part of a synthetic route to certain prostanoids.[3] The reaction required tin tetrachloride and a stoichiometric amount of Wilkinson's catalyst:
An equal amount of a cyclopropane was formed as the result of decarbonylation.
The first catalytic application involved cyclization of 4-pentenal to cyclopentanone using (again) Wilkinson's catalyst.[4] In this reaction the solvent was saturated with ethylene.
- CH2=CHCH2CH2CHO → (CH2)4CO
Reaction mechanism[edit]
Labeling studies establish the following regiochemistry:
- RCDO + CH2=CHR' → RC(O)CH2CHDR'
In terms of the reaction mechanism, hydroacylation begins with oxidative addition of the aldehydic carbon-hydrogen bond. The resulting acyl hydride complex next binds the alkene. The sequence of oxidative addition and alkene coordination is often unclear. Via migratory insertion, the alkene inserts into either the metal-acyl or the metal-hydride bonds. In the final step, the resulting alkyl-acyl or beta-ketoalkyl-hydride complex undergoes reductive elimination.[2] A competing side-reaction is decarbonylation of the aldehyde. This process also proceeds via the intermediacy of the acyl metal hydride:
- R"C(O)-MLn-H → R"-M(CO)Ln-H
This step can be followed by reductive elimination of the alkane:
Asymmetric hydroacylation[edit]
Hydroacylation as an asymmetric reaction was demonstrated in the form of a kinetic resolution.[5][6] A true asymmetric synthesis was also described.[7][8] Both conversions employed rhodium catalysts and a chiral diphosphine ligand. In one application the ligand is Me-DuPhos:[9]
References[edit]
- ^ a b Smith (2020), March's Organic Chemistry, 8th ed. Rxn. 15-30.
- ^ a b Michael C. Willis (2009). "Transition Metal Catalyzed Alkene and Alkyne Hydroacylation". Chem. Rev. 110 (2): 725–748. doi:10.1021/cr900096x. PMID 19873977.
- ^ K. Sakai; J. Ide; O. Oda; N. Nakamura (1972). "Synthetic studies on prostanoids 1 synthesis of methyl 9-oxoprostanoate". Tetrahedron Letters. 13 (13): 1287–1290. doi:10.1016/S0040-4039(01)84569-X.
- ^ Transition-Metal-Promoted Aldehyde-Alkene Addition Reactions Charles F. Lochow, Roy G. Miller J. Am. Chem. Soc., 1976, 98 (5), pp 1281–1283 doi:10.1021/ja00421a050
- ^ The Asymmetric cyclisation of substituted pent-4-enals by a chiral rhodium phosphine catalyst Brian R. James and Charles G. Young J. Chem. Soc., Chem. Commun., 1983, 1215 - 1216, doi:10.1039/C39830001215
- ^ Catalytic decarbonylation, hydroacylation, and resolution of racemic pent-4-enals using chiral bis(di-tertiary-phosphine) complexes of rhodium(I) Brian R. James, and Charles G. Young Journal of Organometallic Chemistry Volume 285, 1985, Pages 321-332 doi:10.1016/0022-328X(85)87377-0
- ^ Asymmetric cyclization reactions by Rh(I) with chiral ligands Yukari Tauraa, Masakazu Tanakaa, Kazuhisa Funakoshia and Kiyoshi Sakai. Tetrahedron Letters. Volume 30, Issue 46, 1989, Pages 6349-6352 doi:10.1016/S0040-4039(01)93891-2
- ^ Asymmetric cyclization reactions. Cyclization of substituted 4-pentenals into cyclopentanone derivatives by rhodium(I) with chiral ligands Yukari Taura, Masakazu Tanaka, Xiao-Ming Wu, Kazuhisa Funakoshi and Kiyoshi Sakai. Tetrahedron. Volume 47, Issue 27, 1991, Pages 4879-4888 doi:10.1016/S0040-4020(01)80954-6
- ^ Synthesis of D- and L-Carbocyclic Nucleosides via Rhodium-Catalyzed Asymmetric Hydroacylation as the Key Step Patricia Marce, Yolanda Dıaz, M. Isabel Matheu, Sergio Castillon Org. Lett., 2008, 10 (21), pp 4735–4738 doi:10.1021/ol801791g