Tn3 transposon

The Tn3 transposon is a 4957 base pair mobile genetic element, found in prokaryotes. It encodes three proteins:
- β-lactamase, an enzyme that confers resistance to β-lactam antibiotics (and is encoded by the gene bla).
- Tn3 transposase (encoded by gene tnpA)
- Tn3 resolvase (encoded by gene tnpR)
Tn3 is an example of replicative transposon (Copy-Paste type transposone).
Initially discovered as a repressor of transposase, resolvase also plays a role in facilitating Tn3 replication (Sherratt 1989).
The transposon is flanked by a pair of 38bp inverted repeats.
Mechanism of replication
[edit]
Step 1 – Replicative integration
[edit]This first stage is catalysed by transposase.
The plasmid containing the transposon (the donor plasmid) fuses with a host plasmid (the target plasmid). In the process, the transposon and a short section of host DNA are replicated. The end product is a 'cointegrate' plasmid containing two copies of the transposon.
Shapiro (1978)[1] proposed the following mechanism for this process:
- Four single-strand cleavages occur – one on each strand of the donor plasmid and one on each strand of the target plasmid.
- The donor and target plasmids are ligated together, but there are two single-stranded regions, due to the positioning of the original cleavages.
- DNA replication makes the single-stranded regions double stranded, using the existing strand as a template. It is in this stage that the transposon is replicated.
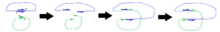
N.B. Diagram is not intended as an accurate representation of 3D structure.
The diagrams on the right illustrate the way in which the positions of the cleavages lead to the replication of certain regions once the plasmids have fused.
Step 2 – Resolution
[edit]
To separate the host and target molecules Tn3 resolvase executes site-specific recombination between the old and new copy of transposon at a specific site called res, which is present in each copy of the transposon. Res is 114 bp long and it consists of 3 sub-sites, namely sites I, II and III. Each of these sites is of different lengths (28, 34 and 25bp, respectively) and they are unevenly spaced with 22bp separating sites I and II and only 5bp between sites II and III. The sites consist of 6bp inverted repeat motifs flanking a central sequence of variable length. These motifs act as binding sites for resolvase, so that each site binds a resolvase dimer but with varying affinity and probably a slightly different protein-DNA complex architecture.[2][3] All three sub-sites are essential for recombination.
At recombination, two directly repeated res sites with resolvase dimers bound to each sub-site, come together to form a large complex structure called the synaptosome. Resolvase bound to sites II and III initiates the assembly of this complex. In this structure, exact architecture of which is still unclear, two res sites are intertwined in such a way as to juxtapose two copies of site I, allowing resolvase dimers bound to each site to form a tetramer. Again, it is the interaction between the resolvase dimers bound at accessory sites (sites II and III) and resolvase at site I that causes the two dimers to synapse and form a tetramer. After the tetramer is formed it becomes activated and the top and bottom DNA strands are simultaneously cleaved in the middle of the site I with a 2bp overhang. The strand exchange ensues by as yet unknown mechanism with a resulting net rotation of 180°. The strand exchange is then followed by the religation (Stark et al., 1992). Recombination between two directly repeated res sites separates, or resolves, the "cointegrate" into two original molecules, each one now containing a copy of the Tn3 transposon. After resolution these two molecules remain linked as a simple two-noded catenane which can be easily separated in vivo by a type II topoisomerase (Grindley 2002). Wild type resolvase system absolutely requires a supercoiled substrate and that the recombination sites are oriented in a direct repeat on the same DNA molecule. However, a number of "deregulated" or "hyperactive" mutants that have lost the requirement for the accessory sites have been isolated. These mutants are capable of catalysing recombination between two copies of site I only, which basically reduces the recombination site size from 114bp to only 28bp.[4][5] Furthermore, these mutants have no supercoiling or connectivity requirements (Arnold et al., 1999) and have been shown to work in mammalian cells.[6] Hyperactive resolvase mutants have so far proven useful in creating resolvases with altered sequence specificity[7] but also in structural work.[8]
The entire resolvase recombination reaction can be reproduced in vitro, requiring only resolvase, a substrate DNA and multivalent cations, using either wild type protein or hyperactive mutants.[4][9]
Hyperactive resolvase mutants, if further developed, could become an alternative to Cre and FLP, the most commonly used recombination systems in molecular biology to date.
References
[edit]- ^ Shapiro, James (April 1979). "Molecular model for the transposition and replication of bacteriophage Mu and other transposable elements". PNAS. 76 (4): 1933–1937. Bibcode:1979PNAS...76.1933S. doi:10.1073/pnas.76.4.1933. PMC 383507. PMID 287033.
- ^ Abdel-Meguid SS, Grindley ND, Templeton NS, Steitz TA (April 1984). "Cleavage of the site-specific recombination protein gamma delta resolvase: the smaller of two fragments binds DNA specifically". Proc. Natl. Acad. Sci. U.S.A. 81 (7): 2001–5. Bibcode:1984PNAS...81.2001A. doi:10.1073/pnas.81.7.2001. PMC 345424. PMID 6326096.
- ^ Blake DG, Boocock MR, Sherratt DJ, Stark WM (September 1995). "Cooperative binding of Tn3 resolvase monomers to a functionally asymmetric binding site". Curr. Biol. 5 (9): 1036–46. doi:10.1016/S0960-9822(95)00208-9. PMID 8542280.
- ^ a b Arnold PH, Blake DG, Grindley ND, Boocock MR, Stark WM (March 1999). "Mutants of Tn3 resolvase which do not require accessory binding sites for recombination activity". EMBO J. 18 (5): 1407–14. doi:10.1093/emboj/18.5.1407. PMC 1171230. PMID 10064606.
- ^ Burke ME, Arnold PH, He J, et al. (February 2004). "Activating mutations of Tn3 resolvase marking interfaces important in recombination catalysis and its regulation". Mol. Microbiol. 51 (4): 937–48. doi:10.1046/j.1365-2958.2003.03831.x. PMID 14763971.
- ^ Schwikardi M, Dröge P (April 2000). "Site-specific recombination in mammalian cells catalyzed by gammadelta resolvase mutants: implications for the topology of episomal DNA". FEBS Lett. 471 (2–3): 147–50. doi:10.1016/S0014-5793(00)01394-6. PMID 10767411. S2CID 83750793.
- ^ Akopian A, He J, Boocock MR, Stark WM (July 2003). "Chimeric recombinases with designed DNA sequence recognition". Proc. Natl. Acad. Sci. U.S.A. 100 (15): 8688–91. Bibcode:2003PNAS..100.8688A. doi:10.1073/pnas.1533177100. PMC 166373. PMID 12837939.
- ^ Li W, Kamtekar S, Xiong Y, Sarkis GJ, Grindley ND, Steitz TA (August 2005). "Structure of a synaptic gammadelta resolvase tetramer covalently linked to two cleaved DNAs". Science. 309 (5738): 1210–5. Bibcode:2005Sci...309.1210L. doi:10.1126/science.1112064. PMID 15994378. S2CID 84409916.
- ^ Reed RR, Grindley ND (September 1981). "Transposon-mediated site-specific recombination in vitro: DNA cleavage and protein-DNA linkage at the recombination site". Cell. 25 (3): 721–8. doi:10.1016/0092-8674(81)90179-3. PMID 6269756. S2CID 28410571.
- Sherratt, D. J. (1989). Tn3 and related transposable elements: site-specific recombination and transposition. In Berg, D. E., Howe, M. (eds) Mobile DNA. American Society for Microbiology, Washinghton, DC pp. 163–184
- Grindley, N.D.F. (2002). The movement of Tn3-like elements: transposition and cointegrate resolution. In Mobile DNA II, Craig, N., Craigie, R., Gellert, M. and Lambowitz, A. (ed.), pp272–302. ASM Press, Washington, DC, USA
