Tumor microenvironment


The tumor microenvironment is a complex ecosystem surrounding a tumor, composed of cancer cells, stromal tissue (including blood vessels, immune cells, fibroblasts and signaling molecules) and the extracellular matrix.[2][3][4][5] Mutual interaction between cancer cells and the different components of the tumor microenvironment support its growth and invasion in healthy tissues which correlates with tumor resistance to current treatments and poor prognosis. The tumor microenvironment is in constant change because of the tumor's ability to influence the microenvironment by releasing extracellular signals, promoting tumor angiogenesis and inducing peripheral immune tolerance, while the immune cells in the microenvironment can affect the growth and evolution of cancerous cells.[2][6][7][8][9]
History
[edit]The concept of the tumor microenvironment (TME) dates back to 1863 when Rudolf Virchow established a connection between inflammation and cancer. However, it was not until 1889 that Stephen Paget's "seed and soil" theory introduced the important role of TME in cancer metastasis, highlighting the intricate relationship between tumors and their surrounding microenvironment. The theory indicated that cancer cells have tendencies when spreading. Paget proposed that the metastases of a particular type of cancer ("the seed") often metastasizes to certain sites ("the soil") based on the similarity of the original and secondary tumor sites. In other words, just as seeds need fertile soil to grow, cancer cells require a supportive microenvironment to metastasize.[10][11]
In 1928, James Ewing challenged Paget's theory with his own perspective on cancer metastasis. Ewing proposed that the ability of cancer cells to metastasize was primarily influenced by mechanical mechanisms such as anatomical and hemodynamic factors of the vascular connection, with tumor cells more likely to be trapped in the first connected organ.[10][12] This viewpoint suggested that certain properties or mutations within cancer cells might dictate their metastatic potential, independent of the surrounding tissue environment.[10] Isaiah Fidler formulated a complementary hypothesis in the 1970s, where he proposed that while the mechanical aspects of blood flow is important, metastatic colonization specifically targets certain organs, known as organotropism.[13]
In the late 1970s, attention shifted towards understanding the role of lymphocytes within the tumor microenvironment. Reports emerged detailing the presence and activities of tumor-infiltrating T and B lymphocytes, as well as natural killer (NK) cells. Researchers observed that tumor-infiltrating T cells had both anti-tumor cytotoxicity and immune-suppressive properties. However, their cytotoxic activity was found to be lower compared to lymphocytes from distant sites, likely due to the overall immunosuppressive state in tumor-bearing individuals.[14]
Vasculature
[edit]A tumor's vasculature is important to its growth, as blood vessels deliver oxygen, nutrients, and growth factors to the tumor.[15] Tumors smaller than 1–2 mm in diameter are delivered oxygen and nutrients through passive diffusion. In larger tumors the center becomes too far away from the existing blood supply, leading the tumor microenvironment to become hypoxic and acidic.[16] Angiogenesis is upregulated to feed the cancer cells and is linked to tumor malignancy.[17]
Endothelial cells and angiogenesis
[edit]In hypoxic environments the tissue sends out signals called hypoxia inducible factors (HIFs) that can stimulate nearby endothelial cells to secrete factors such as vascular endothelial growth factor (VEGF). VEGF activates the endothelial cells, which begins the process of angiogenesis, where new blood vessels emerge from pre-existing vasculature.[18] The blood vessel formed in the tumor environment often does not mature properly, and as a result the vasculature formed in the tumor microenvironment differs from that of normal tissue. The blood vessels formed are often "leaky" and tortuous, with a compromised blood flow.[19][16] As tumors cannot grow large without proper vasculature, sustained angiogenesis is therefore considered one of the hallmarks of cancer.[20]
In later stages of tumor progression endothelial cells can differentiate into carcinoma associated fibroblasts, which furthers metastasis.[16]
Enhanced permeability and retention effect
[edit]The enhanced permeability and retention effect is the observation that the vasculature of tumors tend to accumulate macromolecules in the blood stream to a greater extent than in normal tissue. This is due to the "leaky" nature of the vasculature around tumors, and a lacking lymphatic system.[21] The permeable vasculature allows for easier delivery of therapeutic drugs to the tumor, and the lacking lymphatic vessels contribute to an increased retention. The permeable vasculature is thought to have several causes, including insufficient pericytes and a malformed basement membrane.[22]
Hypoxia
[edit]
While angiogenesis can reduce the hypoxia in the tumor microenvironment, the partial pressure of oxygen is below 5 mmHg in over 50% of locally advanced solid tumors, compared to venous blood which has a partial pressure of oxygen at 40-60 mmHg.[18][5] A hypoxic environment leads to genetic instability by downregulating genes involved in DNA repair mechanisms such as nucleotide excision repair and mismatch repair pathways.[23] This genetic instability leads to a high number of mutated cells, and is associated with cancer progression.[5] Periods of mild and acute hypoxia and reoxygenation can lead cancer cells to adapt and grow into more aggressive phenotypes.[18]
Hypoxia causes the upregulation of hypoxia induced factors (HIFs), which are transcription factors that decides how cells respond to a lack of oxygen.[16] HIFs induces the transcription of thousands of genes, some of which induces angiogenesis or furthers metastasis, leading, for instance, to increased cell migration and matrix remodeling.[24][4] An increased HIF expression can lead tumor cells to shift their metabolism from aerobic to anaerobic, where they obtain energy through glycolysis.[25] Cells with an elevated glucose metabolism produce lactate, which decreases the pH in the microenvironment from a neutral and healthy 7.35-7.45 to an acidic 6.3-7.0. This phenomenon is described as the "Warburg effect".[25][26] HIFs also regulate immune cells, and an increased expression can lead to the inactivation of anti-tumor functions. This furthers the survival of tumor cells and hinders anti-tumor treatment.[25]
Stromal cells
[edit]Cancer is a complex disease involving both tumor cells and surrounding stromal cells. In cancer biology, the stroma is defined as the nonmalignant cells found in the supportive tissue surrounding tumors. These cells include fibroblasts, immune cells, endothelial cells, and various other cell types.[27]
Stromal cells within the tumor microenvironment represent an important cellular component in cancer development, influencing tumor metabolism, growth, metastasis, immune evasion, and resistance to chemotherapy. These cells can originate from neighboring non-cancerous stromal cells or undergo transdifferentiation from tumor cells. Stromal cells contribute to tumor initiation, progression and drug resistance, and the stroma is known to evolve as the tumor develops. Understanding the interactions between cancer cells and stromal cells is essential for developing effective cancer treatments.[27][28] Alterations in the stroma, including the activation of fibroblasts into carcinoma-associated fibroblasts (CAFs) and remodeling of the extracellular matrix (ECM), are recognized as important in cancer progression and potential targets for therapy and diagnosis.[29]
Carcinoma-associated fibroblasts
[edit]
Carcinoma-associated fibroblasts (CAFs) are a heterogenous group of activated fibroblasts central to the reactive stroma within the tumor microenvironment. The precise definition of CAFs remains challenging due to variations in cellular origins and expression markers. However, evidence suggests CAFs originate from activated resident fibroblasts, bone marrow-derived mesenchymal stem cells (MSCs), cancer cells undergoing epithelial-mesenchymal transition (ETM), or endothelial cells through endothelial to mesenchymal transition (EndMT).[31][32][11]
CAFs are one of the most common components of the tumor stroma and are particularly found in the interstitial spaces of breast, prostate, and pancreatic cancer.[28] They interact with cancer cells by secreting a variety of extracellular matrix components or cell-cell adhesion, which is important in regulating the biological behavior of tumors. These regulations are particularly important for tumor development and influence cancer cell growth, invasion, inflammation, and angiogenesis. CAFs may also exhibit tumor-inhibitory properties in some cases.[28][33][11]
CAFs play a dual role in tumorigenesis; one that promotes tumor growth and another that inhibits it, with the former being more common and contributing to tumor development and therapy resistance through various mechanisms.[11][33] Various subpopulations of CAFs have been identified across different cancer types. In breast cancer, for example, studies using single-cell RNA sequencing have revealed distinct phenotypes, including vascular CAFs, matrix CAFs, cycling CAFs, and developmental CAFs.[34] Studies using proteomic analysis and single-cell RNA sequencing have shed more light on the diverse characteristics of CAFs, revealing distinct and sometimes contradictory functions. Their functions appear to be context dependent.[31] This diversity in stomal composition not only shapes the tumor microenvironment, but also affects the behavior of tumor cells.[34][35]
Targeting CAF has emerged as a promising strategy for improving cancer treatment, but the research faces several challenges. These include gaps in our understanding of CAF origins and their diverse functions, some of which may be helpful in combating tumors.[11][33][28]
Extracellular matrix remodeling
[edit]
The extracellular matrix (ECM) is a tree-dimensional network of proteins and proteoglycans in the microenvironment and is present in all tissue. The ECM is a highly dynamic structure and is essential for tissue development, repair, support, and homeostasis.[36][37] In healthy skin, the EMC is composed of various molecules such as collagens, glycoproteins, and glycosaminoglycans that regulate functions and mechanical properties. However, in tumors, the ECM plays an important role in shaping the tumor microenvironment and influences cancer progression, metastasis, and therapeutic response. This process is called extracellular matrix remodeling and is characterized by changes in protein content and enzymatic activity which influences signal transduction and cell-matrix alterations.[38] ECM remodeling involves dynamic alterations in ECM composition, organization, and biomechanical properties. ECM remodeling is induced by factors such as hypoxia, acidosis, inflammatory cells, or proteases secreted by tumor or stromal cells.[39]
Cellular mechanisms
[edit]Cells interact with and bind to the ECM through transmembrane receptors like integrins, discoidin domain receptor 2 (DDRs), and syndecans. The transmission of signals from the ECM to the cell interior involves various pathways. One primary way is direct transduction mediated by transmembrane proteins like integrins.[40] Integrins is the most studied ECM binding receptor and mediate ECM remodeling and regular cellular processes like proliferation, survival, migration, and invasion in response to ECM changes. They act as mechanotransducers by converting mechanical forces from the ECM or the cytoskeleton into chemical signals. Integrins can sense differences between simple, rigid two-dimensional surfaces and complex, malleable three-dimensional environments, altering cellular signaling accordingly.[41][42]
In addition to integrins, other cell receptors like cell surface glycoprotein receptor (CD44), DDR2 and elastin-binding protein receptor (EBPR) can activate signaling pathways such as phosphatidylinositol 3-kinase and Akt. These receptors interact with various ECM components and create diverse cellular processes that contribute both to normal physiological functions and pathological conditions like cancer.[40]
Impact on cancer progression
[edit]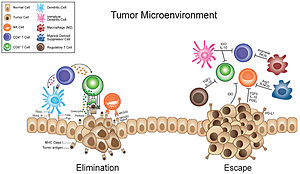
While ECM remodeling is tightly regulated under normal physiological conditions, it also modulates many of the tumor cell behaviors associated with cancer progression. This includes evasion of apoptosis, sustained angiogenesis, limitless replication potential, and tissue invasion.[43][44] In cancer, changes in the ECM dynamics lead to changes in composition, density, and mechanical properties, affecting tumor aggressiveness and response to therapy. Research suggests that both pro- and anti-tumorigenic effects occurs during ECM remodeling. In early tumor formation, stromal cells produce excess ECM proteins, causing the tissue around the tumor to stiffen. Some of the contributing factors to tumor stiffness is increased type 1 collagen and acid deposition.[43][45] Additionally, the restructured ECM and its degradation fragments (matrikines) impacts signaling pathways via cell-surface receptor interactions, leading to dysregulated stromal cell behavior and the emergence of an oncogenic microenvironment.[46]
Immune cells
[edit]Tumor-associated immune cells can be tumor-antagonizing or tumor-promoting, meaning that they can suppress or promote tumor growth.[47] Because of the effects of hypoxia, the anti-tumor abilities of many tumor-antagonizing immune cells, such as cytotoxic T cells and natural killer cells, become inhibited. Tumor-promoting immune cells such as regulatory T cells and myeloid derived suppressor cells will, on the other hand, become upregulated.[25]



Myeloid-derived suppressor cells and tumor-associated macrophages
[edit]Myeloid-derived suppressor cells are a heterogeneous population of cells of myelogenous origin[48] that are considered tumor promoting.[47] They have the potential to repress T cell responses,[49] can support angiogenesis by producing proteins such as vascular endothelial growth factor (VEGF), and can promote metastasis. Tumor associated macrophages with the M2 phenotype are considered myeloid-derived suppressor cells.[47]
Tumor-associated macrophages are a central component in the strong link between chronic inflammation and cancer, and are recruited to the tumor as a response to cancer-associated inflammation.[50] Their sluggish NF-κB activation allows for the smoldering inflammation seen in cancer.[51] Unlike normal macrophages, tumor-associated macrophages lack cytotoxic activity.[52] Monocyte derived macrophages are divided into inflammatory M1-polarized macrophages and anti-inflammatory M2-polarized macrophages. M1-polarized macrophages phagocytize tumor cells and are considered tumor-antagonizing.[16] M2-polarized macrophages are, on the other hand, tumor-promoting, because they promote tumor progression by suppressing immunosurveillance,[47] aiding angiogenesis by secreting vascular endothelial growth factor (VEGF)[5] and remodeling the extracellular matrix.[47] The tumor microenvironment promotes the M2-polarized macrophages, and an increased amount of tumor-associated macrophages is associated with worse prognosis.[16][53][54]
Tumor-associated macrophages are associated with using exosomes to deliver invasion-potentiating microRNA into cancerous cells, specifically breast cancer cells.[49][55]
Neutrophils
[edit]Neutrophils are polymorphonuclear immune cells that are critical components of the innate immune system. Neutrophils can accumulate in tumors and in some cancers, such as lung adenocarcinoma, their abundance at the tumor site is associated with worsened disease prognosis.[56][57][58] Neutrophil numbers (and myeloid cell precursors) in the blood can be increased in some patients with solid tumors.[59][60][61]
Experiments in mice have mainly shown that tumor-associated neutrophils exhibit tumor-promoting functions,[62][63][64][65] but a smaller number of studies show that neutrophils can also inhibit tumor growth.[66][67] Tumor associated neutrophils can be divided into N1- and N2-polarized neutrophils.[47] N1-polarized neutrophils accumulate in the tumor in its early stages and support with tumor cell death. In later stages N2-polarized neutrophils promotes angiogenesis by secreting vascular endothelial growth factor (VEGF).[16]
Tumor infiltrating lymphocytes
[edit]Tumor-infiltrating lymphocytes are lymphocytes, including T cells, B cells and natural killer cells, that penetrate the tumor and have the ability to recognize and kill cancer cells.[68] A high concentration is generally positively correlated with good prognosis (802).[69] This type of immune cells can also block metastasis, as natural killer cells are most efficient at killing cancer cells outside of the tumor microenvironment.[16] Tumor-infiltrating lymphocytes have been used in therapeutic treatments, where lab-amplificated immune cells are transferred to cancer patients to help their immune system fight the cancer.[69] This treatment has seen success in solid tumors such as melanoma.[70]
Tumor-infiltrating lymphocytes can become tumor-promoting due to the immunosuppressive mechanisms of the tumor microenvironment.[69] Cancer cells induce apoptosis of activated T cells by secreting exosomes containing death ligands such as FasL and TRAIL, and via the same method, turn off the normal cytotoxic response of natural killer cells.[71][72]
T cells
[edit]There are several types of T cells that are important to tumorigenesis, including cytotoxic T cells (CD8+), T helper 1 (Th-1) cells and regulatory T cells (Tregs).[16] CD8+ cells are tumor-antagonizing cells that recognize tumor antigens and targets cancer cells for destruction. In addition, CD8+ cells slow tumor progression and suppress angiogenesis by releasing interferon-gamma (IFN-γ).[16] Th-1 cells supports the activation and proliferation of CD8+ cells by secreting IFN-γ and interleukin-2 (IL-2), and by cross-presenting tumor antigens.[47] Tregs are, as opposed to CD8+, tumor promoting. They secrete tumor growth factors, and indirectly support cancer survival by interacting with endothelial cells and carcinoma associated fibroblasts.[16] Tregs also have immunosuppressive mechanisms that can make CD8+ cells less effective.[47]
T cells reach tumor sites via the vascular system, where the tumor microenvironment appears to preferentially recruit other immune cells over T cells. One such discriminating mechanism is the release of cell-type specific chemokines. Another is the expression of the apoptosis inducer Fas ligand (FasL) in the vasculature of ovarian, colon, prostate, breast, bladder and renal tumors. Tumors with a high expression of FasL has been shown to contain an abundancy of Tregs, but few CD8+ cells.[3]
T cells must replicate after arriving at the tumor site to effectively kill the cancer cells, survive hostile elements and migrate through the stroma to the cancer cells. This is affected by the tumor microenvironment. The draining lymph nodes are the likely location for cancer specific T cell replication, although this also occurs within the tumor.[3]
Research
[edit]Models
[edit]Several in vitro and in vivo models have been developed that seek to replicate the TME in a controlled environment. Tumor immortalised cell lines and primary cell cultures have been long used in order to study various tumors. They are quick to set up and inexpensive, but simplistic and prone to genetic drift.[73] 3D tumor models have been developed as a more spatially representative model of the TME. Spheroid cultures, scaffolds and organoids are generally derived from stem cells or ex vivo and are much better at recreating the tumour architecture than 2D cell cultures.[74]
Human germline genetic variants and tumor microenvironment
[edit]Recent research has demonstrated that human germline genetic variants can significantly influence the composition of the tumor microenvironment. These germline variants affect the number of infiltrating CD8 T cells and regulatory T cells within tumors, thereby impacting immune evasion and responses to immunotherapy. Notably, studies published in the Journal of Clinical Investigation[75] and Nature Communications[76] have highlighted the role of STAT3-enhancing germline mutations and other common genetic variants in modulating the tumor immune landscape and driving therapeutic outcomes.
Drug development
[edit]Advancements in remodeling nanotherapeutics have led to progress in suppressing cancer metastasis and reducing the likelihood of cancer occurrence. Strategies included regulation of hypoxia, angiogenesis, cancer-associated fibroblasts (CAFs), extracellular matrix (ECM), and tumor-associated macrophages. These approaches aimed to improve anti-tumor effects and sensitize other therapies.[77] Researchers have discovered that the use of ferumoxytol suppresses tumor growth by inducing transition of macrophages to proinflammatory types.[78] Nanocarrier vehicles (~20–200 nm in diameter) can transport drugs and other therapeutic molecules. These therapies can be targeted to selectively extravasate through tumor vasculature.[22][79] These efforts include protein capsids[80] and liposomes.[81] However, as some important, normal tissues, such as the liver and kidneys, also have fenestrated endothelium, the nanocarrier size (10–100 nm, with greater retention in tumors seen in using larger nanocarriers) and charge (anionic or neutral) must be considered.[22] Lymphatic vessels do not usually develop with the tumor, leading to increased interstitial fluid pressure, which may block tumor access.[22][82]
Therapies
[edit]Antibodies
[edit]Bevacizumab is clinically approved in the US to treat a variety of cancers by targeting VEGF-A, which is produced by both carcinoma associated fibroblasts and tumor-associated macrophages, thus slowing angiogenesis. It was initially approved for metastatic colorectal cancer, but its uses now span various cancers.[83]
Targeting immunoregulatory membrane receptors succeeded in some patients with melanoma, non-small-cell lung carcinoma, urothelial bladder cancer and renal cell cancer. In mice, anti-CTLA-4 therapy leads to clearance from the tumor of FOXP3+ regulatory T cells (Tregs) whose presence may impair effector T cell function.[84]
Kinase inhibitors
[edit]Mutated kinases are common in cancer cells, making them attractive targets for anticancer drugs. Kinase inhibitors are potent, specific and target abnormal kinases while minimizing toxicity. Kinase inhibitors have expanded treatment options for various cancers.[85]
Tyrosine kinase inhibitors (TKIs), such as erlotinib, lapatinib, and gefitinib, target epidermal growth factor receptors (EGFRs) in cancer by blocking the activity of protein tyrosine kinases (PTKs). This show promise in modulating the tumor microenvironment, resulting in cancer regression. Understanding how TKIs modulates the tumor microenvironment may offer another form of cancer treatment.[86][87]
Chimeric antigen receptor cell therapy
[edit]Chimeric antigen receptors (CAR) T cell therapy is an immunotherapy treatment that uses genetically modified T lymphocytes to effectively target tumor cells. CARs are programmed to target tumor-associated antigens as well as replicate rapidly and homogenously, making them potentially very effective as a cancer-therapy.[88][89] Since the tumor microenvironment has several barriers that limit the ability of CAR T cells to infiltrate the tumor, several strategies have been developed to address this. Localized delivery of CAR T cells in glioblastoma suggested improved anti-tumor activity and engineering these cells to overexpress chemokine receptors suggested improvement of CAR T cell trafficking.[90] As this therapy expands to other diseases, managing its unique toxicity profile, including cytokine release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS), and cytopenias, becomes increasingly more important.[91]
See also
[edit]References
[edit]- ^ Piñeiro Fernández J, Luddy KA, Harmon C, O'Farrelly C (2019). "Hepatic Tumor Microenvironments and Effects on NK Cell Phenotype and Function". International Journal of Molecular Sciences. 20 (17): 4131. doi:10.3390/ijms20174131. ISSN 1422-0067. PMC 6747260. PMID 31450598.
- ^ a b Alfarouk KO, Muddathir AK, Shayoub ME (January 2011). "Tumor acidity as evolutionary spite". Cancers. 3 (1): 408–414. doi:10.3390/cancers3010408. PMC 3756368. PMID 24310355.
- ^ a b c Joyce JA, Fearon DT (April 2015). "T cell exclusion, immune privilege, and the tumor microenvironment". Science. 348 (6230): 74–80. Bibcode:2015Sci...348...74J. doi:10.1126/science.aaa6204. PMID 25838376.
- ^ a b Spill F, Reynolds DS, Kamm RD, Zaman MH (August 2016). "Impact of the physical microenvironment on tumor progression and metastasis". Current Opinion in Biotechnology. 40: 41–48. doi:10.1016/j.copbio.2016.02.007. PMC 4975620. PMID 26938687.
- ^ a b c d Weber CE, Kuo PC (September 2012). "The tumor microenvironment". Surgical Oncology. 21 (3): 172–177. doi:10.1016/j.suronc.2011.09.001. PMID 21963199.
- ^ Korneev KV, Atretkhany KN, Drutskaya MS, Grivennikov SI, Kuprash DV, Nedospasov SA (January 2017). "TLR-signaling and proinflammatory cytokines as drivers of tumorigenesis". Cytokine. 89: 127–135. doi:10.1016/j.cyto.2016.01.021. PMID 26854213.
- ^ Ghoshdastider U, Rohatgi N, Mojtabavi Naeini M, Baruah P, Revkov E, Guo YA, et al. (April 2021). "Pan-Cancer Analysis of Ligand-Receptor Cross-talk in the Tumor Microenvironment". Cancer Research. 81 (7): 1802–1812. doi:10.1158/0008-5472.CAN-20-2352. PMID 33547160. S2CID 232432582.
- ^ Žavbi J, Breznik B (2021). "Modelling the microenvironment of the most aggressive brain tumours for preclinical studies". Advances in Cancer Biology - Metastasis. 3: 100017. doi:10.1016/j.adcanc.2021.100017. ISSN 2667-3940. S2CID 244452599.
- ^ Xinyuan T, Lei Y, Jianping S, Rongwei Z, Ruiwen S, Ye Z, et al. (2023-10-01). "Advances in the role of gut microbiota in the regulation of the tumor microenvironment (Review)". Oncology Reports. 50 (4): 1–15. doi:10.3892/or.2023.8618. ISSN 1021-335X. PMC 10485805. PMID 37615187.
- ^ a b c Akhtar M, Haider A, Rashid S, Al-Nabet AD (January 2019). "Paget's "Seed and Soil" Theory of Cancer Metastasis: An Idea Whose Time has Come". Advances in Anatomic Pathology. 26 (1): 69–74. doi:10.1097/PAP.0000000000000219. ISSN 1072-4109. PMID 30339548. S2CID 53010974.
- ^ a b c d e Mao Y, Keller ET, Garfield DH, Shen K, Wang J (2013-06-01). "Stromal cells in tumor microenvironment and breast cancer". Cancer and Metastasis Reviews. 32 (1): 303–315. doi:10.1007/s10555-012-9415-3. ISSN 1573-7233. PMC 4432936. PMID 23114846.
- ^ Liu Q, Zhang H, Jiang X, Qian C, Liu Z, Luo D (2017-12-02). "Factors involved in cancer metastasis: a better understanding to "seed and soil" hypothesis". Molecular Cancer. 16 (1): 176. doi:10.1186/s12943-017-0742-4. ISSN 1476-4598. PMC 5712107. PMID 29197379.
- ^ da Costa VR, Araldi RP, Vigerelli H, D'Ámelio F, Mendes TB, Gonzaga V, et al. (October 2021). "Exosomes in the Tumor Microenvironment: From Biology to Clinical Applications". Cells. 10 (10): 2617. doi:10.3390/cells10102617. ISSN 2073-4409. PMC 8533895. PMID 34685596.
- ^ Maman S, Witz IP (June 2018). "A history of exploring cancer in context". Nature Reviews Cancer. 18 (6): 359–376. doi:10.1038/s41568-018-0006-7. ISSN 1474-1768. PMID 29700396. S2CID 13777357.
- ^ Al-Ostoot FH, Salah S, Khamees HA, Khanum SA (2021-01-01). "Tumor angiogenesis: Current challenges and therapeutic opportunities". Cancer Treatment and Research Communications. 28: 100422. doi:10.1016/j.ctarc.2021.100422. ISSN 2468-2942. PMID 34147821.
- ^ a b c d e f g h i j k Anderson NM, Simon MC (August 2020). "The tumor microenvironment". Current Biology. 30 (16): R921–R925. Bibcode:2020CBio...30.R921A. doi:10.1016/j.cub.2020.06.081. ISSN 0960-9822. PMC 8194051. PMID 32810447.
- ^ Jiang X, Wang J, Deng X, Xiong F, Zhang S, Gong Z, et al. (2020-09-30). "The role of microenvironment in tumor angiogenesis". Journal of Experimental & Clinical Cancer Research. 39 (1): 204. doi:10.1186/s13046-020-01709-5. ISSN 1756-9966. PMC 7526376. PMID 32993787.
- ^ a b c Li Y, Zhao L, Li XF (2021-08-05). "Hypoxia and the Tumor Microenvironment". Technology in Cancer Research & Treatment. 20: 15330338211036304. doi:10.1177/15330338211036304. ISSN 1533-0346. PMC 8358492. PMID 34350796.
- ^ Bhat SM, Badiger VA, Vasishta S, Chakraborty J, Prasad S, Ghosh S, et al. (2021-12-01). "3D tumor angiogenesis models: recent advances and challenges". Journal of Cancer Research and Clinical Oncology. 147 (12): 3477–3494. doi:10.1007/s00432-021-03814-0. ISSN 1432-1335. PMC 8557138. PMID 34613483.
- ^ Hanahan D, Monje M (2023-03-13). "Cancer hallmarks intersect with neuroscience in the tumor microenvironment". Cancer Cell. 41 (3): 573–580. doi:10.1016/j.ccell.2023.02.012. ISSN 1535-6108. PMC 10202656. PMID 36917953.
- ^ Alasvand N, Urbanska AM, Rahmati M, Saeidifar M, Gungor-Ozkerim PS, Sefat F, et al. (2017-01-01), Grumezescu AM (ed.), "Chapter 13 - Therapeutic Nanoparticles for Targeted Delivery of Anticancer Drugs", Multifunctional Systems for Combined Delivery, Biosensing and Diagnostics, Elsevier, pp. 245–259, doi:10.1016/b978-0-323-52725-5.00013-7, ISBN 978-0-323-52725-5, retrieved 2024-02-19
- ^ a b c d Danhier F, Feron O, Préat V (December 2010). "To exploit the tumor microenvironment: Passive and active tumor targeting of nanocarriers for anti-cancer drug delivery". Journal of Controlled Release. 148 (2): 135–146. doi:10.1016/j.jconrel.2010.08.027. PMID 20797419.
- ^ Tang M, Bolderson E, O'Byrne KJ, Richard DJ (2021). "Tumor Hypoxia Drives Genomic Instability". Frontiers in Cell and Developmental Biology. 9. doi:10.3389/fcell.2021.626229. ISSN 2296-634X. PMC 8007910. PMID 33796526.
- ^ Wicks EE, Semenza GL (2022-06-01). "Hypoxia-inducible factors: cancer progression and clinical translation". The Journal of Clinical Investigation. 132 (11). doi:10.1172/JCI159839. ISSN 0021-9738. PMC 9151701. PMID 35642641.
- ^ a b c d Chen G, Wu K, Li H, Xia D, He T (2022). "Role of hypoxia in the tumor microenvironment and targeted therapy". Frontiers in Oncology. 12. doi:10.3389/fonc.2022.961637. ISSN 2234-943X. PMC 9545774. PMID 36212414.
- ^ Lee SH, Griffiths JR (June 2020). "How and Why Are Cancers Acidic? Carbonic Anhydrase IX and the Homeostatic Control of Tumour Extracellular pH". Cancers. 12 (6): 1616. doi:10.3390/cancers12061616. PMC 7352839. PMID 32570870.
- ^ a b Werb Z, Lu P (July–August 2015). "The Role of Stroma in Tumor Development". The Cancer Journal. 21 (4): 250–253. doi:10.1097/PPO.0000000000000127. ISSN 1540-336X. PMC 4963227. PMID 26222075.
- ^ a b c d Zhao Y, Shen M, Wu L, Yang H, Yao Y, Yang Q, et al. (2023-09-04). "Stromal cells in the tumor microenvironment: accomplices of tumor progression?". Cell Death & Disease. 14 (9): 587. doi:10.1038/s41419-023-06110-6. ISSN 2041-4889. PMC 10477351. PMID 37666813.
- ^ Belhabib I, Zaghdoudi S, Lac C, Bousquet C, Jean C (2021-07-11). "Extracellular Matrices and Cancer-Associated Fibroblasts: Targets for Cancer Diagnosis and Therapy?". Cancers. 13 (14): 3466. doi:10.3390/cancers13143466. ISSN 2072-6694. PMC 8303391. PMID 34298680.
- ^ Arora L, Pal D (2021). "Remodeling of Stromal Cells and Immune Landscape in Microenvironment During Tumor Progression". Frontiers in Oncology. 11. doi:10.3389/fonc.2021.596798. ISSN 2234-943X. PMC 7982455. PMID 33763348.
- ^ a b Glabman RA, Choyke PL, Sato N (January 2022). "Cancer-Associated Fibroblasts: Tumorigenicity and Targeting for Cancer Therapy". Cancers. 14 (16): 3906. doi:10.3390/cancers14163906. ISSN 2072-6694. PMC 9405783. PMID 36010899.
- ^ Yang D, Liu J, Qian H, Zhuang Q (July 2023). "Cancer-associated fibroblasts: from basic science to anticancer therapy". Experimental & Molecular Medicine. 55 (7): 1322–1332. doi:10.1038/s12276-023-01013-0. ISSN 2092-6413. PMC 10394065. PMID 37394578.
- ^ a b c Ping Q, Yan R, Cheng X, Wang W, Zhong Y, Hou Z, et al. (September 2021). "Cancer-associated fibroblasts: overview, progress, challenges, and directions". Cancer Gene Therapy. 28 (9): 984–999. doi:10.1038/s41417-021-00318-4. ISSN 1476-5500. PMID 33712707.
- ^ a b Cords L, Tietscher S, Anzeneder T, Langwieder C, Rees M, de Souza N, et al. (2023-07-18). "Cancer-associated fibroblast classification in single-cell and spatial proteomics data". Nature Communications. 14 (1): 4294. Bibcode:2023NatCo..14.4294C. doi:10.1038/s41467-023-39762-1. ISSN 2041-1723. PMC 10354071. PMID 37463917.
- ^ Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, et al. (March 2020). "A framework for advancing our understanding of cancer-associated fibroblasts". Nature Reviews Cancer. 20 (3): 174–186. doi:10.1038/s41568-019-0238-1. ISSN 1474-1768. PMC 7046529. PMID 31980749.
- ^ Bonnans C, Chou J, Werb Z (2015-02-04). "Remodelling the extracellular matrix in development and disease". Nature Reviews Molecular Cell Biology. 15 (12): 786–801. doi:10.1038/nrm3904. ISSN 1471-0080. PMC 4316204. PMID 25415508.
- ^ Fromme JE, Zigrino P (2022). "The Role of Extracellular Matrix Remodeling in Skin Tumor Progression and Therapeutic Resistance". Frontiers in Molecular Biosciences. 9. doi:10.3389/fmolb.2022.864302. ISSN 2296-889X. PMC 9086898. PMID 35558554.
- ^ Diller RB, Tabor AJ (2022-07-01). "The Role of the Extracellular Matrix (ECM) in Wound Healing: A Review". Biomimetics. 7 (3): 87. doi:10.3390/biomimetics7030087. ISSN 2313-7673. PMC 9326521. PMID 35892357.
- ^ Brassart-Pasco S, Brézillon S, Brassart B, Ramont L, Oudart JB, Monboisse JC (2020). "Tumor Microenvironment: Extracellular Matrix Alterations Influence Tumor Progression". Frontiers in Oncology. 10: 397. doi:10.3389/fonc.2020.00397. ISSN 2234-943X. PMC 7174611. PMID 32351878.
- ^ a b Mohan V, Das A, Sagi I (2020-05-01). "Emerging roles of ECM remodeling processes in cancer". Seminars in Cancer Biology. Translating Extracellular Matrix. 62: 192–200. doi:10.1016/j.semcancer.2019.09.004. ISSN 1044-579X. PMID 31518697. S2CID 202571768.
- ^ Larsen M, Artym VV, Green JA, Yamada KM (2006-10-01). "The matrix reorganized: extracellular matrix remodeling and integrin signaling". Current Opinion in Cell Biology. Cell-to-cell contact and extracellular matrix. 18 (5): 463–471. doi:10.1016/j.ceb.2006.08.009. ISSN 0955-0674. PMID 16919434.
- ^ Kai F, Drain AP, Weaver VM (2019-05-06). "The Extracellular Matrix Modulates the Metastatic Journey". Developmental Cell. 49 (3): 332–346. doi:10.1016/j.devcel.2019.03.026. ISSN 1878-1551. PMC 6527347. PMID 31063753.
- ^ a b Dzobo K, Dandara C (2023-04-05). "The Extracellular Matrix: Its Composition, Function, Remodeling, and Role in Tumorigenesis". Biomimetics. 8 (2): 146. doi:10.3390/biomimetics8020146. ISSN 2313-7673. PMC 10123695. PMID 37092398.
- ^ Giussani M, Triulzi T, Sozzi G, Tagliabue E (2019-01-23). "Tumor Extracellular Matrix Remodeling: New Perspectives as a Circulating Tool in the Diagnosis and Prognosis of Solid Tumors". Cells. 8 (2): 81. doi:10.3390/cells8020081. ISSN 2073-4409. PMC 6406979. PMID 30678058.
- ^ Wullkopf L, West AK, Leijnse N, Cox TR, Madsen CD, Oddershede LB, et al. (October 2018). Discher D (ed.). "Cancer cells' ability to mechanically adjust to extracellular matrix stiffness correlates with their invasive potential". Molecular Biology of the Cell. 29 (20): 2378–2385. doi:10.1091/mbc.E18-05-0319. ISSN 1059-1524. PMC 6233061. PMID 30091653.
- ^ Popova NV, Jücker M (January 2022). "The Functional Role of Extracellular Matrix Proteins in Cancer". Cancers. 14 (1): 238. doi:10.3390/cancers14010238. ISSN 2072-6694. PMC 8750014. PMID 35008401.
- ^ a b c d e f g h Lei X, Lei Y, Li JK, Du WX, Li RG, Yang J, et al. (2020-02-01). "Immune cells within the tumor microenvironment: Biological functions and roles in cancer immunotherapy". Cancer Letters. 470: 126–133. doi:10.1016/j.canlet.2019.11.009. ISSN 0304-3835. PMID 31730903. S2CID 208063582.
- ^ Mantovani A, Allavena P, Sica A, Balkwill F (July 2008). "Cancer-related inflammation". Nature. 454 (7203): 436–444. Bibcode:2008Natur.454..436M. doi:10.1038/nature07205. hdl:2434/145688. PMID 18650914. S2CID 4429118.
- ^ a b Mathias RA, Gopal SK, Simpson RJ (January 2013). "Contribution of cells undergoing epithelial-mesenchymal transition to the tumour microenvironment". Journal of Proteomics. 78: 545–557. doi:10.1016/j.jprot.2012.10.016. PMID 23099347.
- ^ Balkwill F, Charles KA, Mantovani A (March 2005). "Smoldering and polarized inflammation in the initiation and promotion of malignant disease". Cancer Cell. 7 (3): 211–217. doi:10.1016/j.ccr.2005.02.013. PMID 15766659.
- ^ Biswas SK, Gangi L, Paul S, Schioppa T, Saccani A, Sironi M, et al. (March 2006). "A distinct and unique transcriptional program expressed by tumor-associated macrophages (defective NF-kappaB and enhanced IRF-3/STAT1 activation)". Blood. 107 (5): 2112–2122. doi:10.1182/blood-2005-01-0428. PMID 16269622. S2CID 5884781.
- ^ Qian BZ, Pollard JW (April 2010). "Macrophage diversity enhances tumor progression and metastasis". Cell. 141 (1): 39–51. doi:10.1016/j.cell.2010.03.014. PMC 4994190. PMID 20371344.
- ^ Zhang W, Wang L, Zhou D, Cui Q, Zhao D, Wu Y (January 2011). "Expression of tumor-associated macrophages and vascular endothelial growth factor correlates with poor prognosis of peripheral T-cell lymphoma, not otherwise specified". Leukemia & Lymphoma. 52 (1): 46–52. doi:10.3109/10428194.2010.529204. PMID 21077742. S2CID 26116121.
- ^ Zhang BC, Gao J, Wang J, Rao ZG, Wang BC, Gao JF (December 2011). "Tumor-associated macrophages infiltration is associated with peritumoral lymphangiogenesis and poor prognosis in lung adenocarcinoma". Medical Oncology. 28 (4): 1447–1452. doi:10.1007/s12032-010-9638-5. PMID 20676804. S2CID 24840259.
- ^ Yang M, Chen J, Su F, Yu B, Su F, Lin L, et al. (September 2011). "Microvesicles secreted by macrophages shuttle invasion-potentiating microRNAs into breast cancer cells". Molecular Cancer. 10 (117): 117. doi:10.1186/1476-4598-10-117. PMC 3190352. PMID 21939504.
- ^ Coffelt SB, Wellenstein MD, de Visser KE (July 2016). "Neutrophils in cancer: neutral no more" (PDF). Nature Reviews. Cancer. 16 (7): 431–446. doi:10.1038/nrc.2016.52. PMID 27282249. S2CID 4393159.
- ^ Gentles AJ, Newman AM, Liu CL, Bratman SV, Feng W, Kim D, et al. (August 2015). "The prognostic landscape of genes and infiltrating immune cells across human cancers". Nature Medicine. 21 (8): 938–945. doi:10.1038/nm.3909. PMC 4852857. PMID 26193342.
- ^ Engblom C, Pfirschke C, Pittet MJ (July 2016). "The role of myeloid cells in cancer therapies". Nature Reviews. Cancer. 16 (7): 447–462. doi:10.1038/nrc.2016.54. PMID 27339708. S2CID 21924175.
- ^ Huang SH, Waldron JN, Milosevic M, Shen X, Ringash J, Su J, et al. (February 2015). "Prognostic value of pretreatment circulating neutrophils, monocytes, and lymphocytes in oropharyngeal cancer stratified by human papillomavirus status". Cancer. 121 (4): 545–555. doi:10.1002/cncr.29100. PMID 25336438. S2CID 926930.
- ^ Jiang L, Jiang S, Situ D, Lin Y, Yang H, Li Y, et al. (April 2015). "Prognostic value of monocyte and neutrophils to lymphocytes ratio in patients with metastatic soft tissue sarcoma". Oncotarget. 6 (11): 9542–9550. doi:10.18632/oncotarget.3283. PMC 4496237. PMID 25865224.
- ^ Wu WC, Sun HW, Chen HT, Liang J, Yu XJ, Wu C, et al. (March 2014). "Circulating hematopoietic stem and progenitor cells are myeloid-biased in cancer patients". Proceedings of the National Academy of Sciences of the United States of America. 111 (11): 4221–4226. Bibcode:2014PNAS..111.4221W. doi:10.1073/pnas.1320753111. PMC 3964061. PMID 24591638.
- ^ Coffelt SB, Kersten K, Doornebal CW, Weiden J, Vrijland K, Hau CS, et al. (June 2015). "IL-17-producing γδ T cells and neutrophils conspire to promote breast cancer metastasis". Nature. 522 (7556): 345–348. Bibcode:2015Natur.522..345C. doi:10.1038/nature14282. PMC 4475637. PMID 25822788.
- ^ Engblom C, Pfirschke C, Zilionis R, Da Silva Martins J, Bos SA, Courties G, et al. (December 2017). "Osteoblasts remotely supply lung tumors with cancer-promoting SiglecFhigh neutrophils". Science. 358 (6367): eaal5081. doi:10.1126/science.aal5081. PMC 6343476. PMID 29191879.
- ^ Casbon AJ, Reynaud D, Park C, Khuc E, Gan DD, Schepers K, et al. (February 2015). "Invasive breast cancer reprograms early myeloid differentiation in the bone marrow to generate immunosuppressive neutrophils". Proceedings of the National Academy of Sciences of the United States of America. 112 (6): E566–E575. Bibcode:2015PNAS..112E.566C. doi:10.1073/pnas.1424927112. PMC 4330753. PMID 25624500.
- ^ Wculek SK, Malanchi I (December 2015). "Neutrophils support lung colonization of metastasis-initiating breast cancer cells". Nature. 528 (7582): 413–417. Bibcode:2015Natur.528..413W. doi:10.1038/nature16140. PMC 4700594. PMID 26649828.
- ^ Finisguerra V, Di Conza G, Di Matteo M, Serneels J, Costa S, Thompson AA, et al. (June 2015). "MET is required for the recruitment of anti-tumoural neutrophils". Nature. 522 (7556): 349–353. Bibcode:2015Natur.522..349F. doi:10.1038/nature14407. PMC 4594765. PMID 25985180.
- ^ Granot Z, Henke E, Comen EA, King TA, Norton L, Benezra R (September 2011). "Tumor entrained neutrophils inhibit seeding in the premetastatic lung". Cancer Cell. 20 (3): 300–314. doi:10.1016/j.ccr.2011.08.012. PMC 3172582. PMID 21907922.
- ^ "tumor-infiltrating lymphocyte". www.cancer.gov. 2011-02-02. Retrieved 2024-02-19.
- ^ a b c Whiteside TL (2022). "Tumor-Infiltrating Lymphocytes and Their Role in Solid Tumor Progression". Interaction of Immune and Cancer Cells. Experientia Supplementum. Vol. 113. pp. 89–106. doi:10.1007/978-3-030-91311-3_3. ISBN 978-3-030-91310-6. ISSN 1664-431X. PMC 9113058. PMID 35165861.
- ^ Turcotte S, Rosenberg SA (2011). "Immunotherapy for metastatic solid cancers". Advances in Surgery. 45: 341–360. doi:10.1016/j.yasu.2011.04.003. PMC 3578602. PMID 21954698.
- ^ Valenti R, Huber V, Iero M, Filipazzi P, Parmiani G, Rivoltini L (April 2007). "Tumor-released microvesicles as vehicles of immunosuppression". Cancer Research. 67 (7): 2912–2915. doi:10.1158/0008-5472.CAN-07-0520. PMID 17409393.
- ^ Clayton A, Tabi Z (May–June 2005). "Exosomes and the MICA-NKG2D system in cancer". Blood Cells, Molecules & Diseases. 34 (3): 206–213. doi:10.1016/j.bcmd.2005.03.003. PMID 15885603.
- ^ Freedman LP, Gibson MC, Ethier SP, Soule HR, Neve RM, Reid YA (June 2015). "Reproducibility: changing the policies and culture of cell line authentication". Nature Methods. 12 (6): 493–497. doi:10.1038/nmeth.3403. PMID 26020501. S2CID 20557369.
- ^ Franchi-Mendes T, Eduardo R, Domenici G, Brito C (September 2021). "3D Cancer Models: Depicting Cellular Crosstalk within the Tumour Microenvironment". Cancers. 13 (18): 4610. doi:10.3390/cancers13184610. PMC 8468887. PMID 34572836.
- ^ Kogan D, Grabner A, Yanucil C, Faul C, Ulaganathan VK (1 May 2018). "STAT3-enhancing germline mutations contribute to tumor-extrinsic immune evasion". Journal of Clinical Investigation. 128 (5): 1867–1872. doi:10.1172/JCI96708. PMC 5919827.
- ^ Pagadala M, Sears TJ, Wu VH, Pérez-Guijarro E, Kim H, Castro A, et al. (12 May 2023). "Germline modifiers of the tumor immune microenvironment implicate drivers of cancer risk and immunotherapy response". Nature Communications. 14 (1). doi:10.1038/s41467-023-38271-5. PMC 10182072.
- ^ Feng Y, Liao Z, Zhang H, Xie X, You F, Liao X, et al. (January 2023). "Emerging nanomedicines strategies focused on tumor microenvironment against cancer recurrence and metastasis". Chemical Engineering Journal. 452: 139506. Bibcode:2023ChEnJ.45239506F. doi:10.1016/j.cej.2022.139506. S2CID 252676223.
- ^ Raju GS, Pavitra E, Varaprasad GL, Bandaru SS, Nagaraju GP, Farran B, et al. (June 2022). "Nanoparticles mediated tumor microenvironment modulation: current advances and applications". Journal of Nanobiotechnology. 20 (1): 274. doi:10.1186/s12951-022-01476-9. PMC 9195263. PMID 35701781.
- ^ Unezaki S, Maruyama K, Hosoda JI, Nagae I, Koyanagi Y, Nakata M, et al. (22 November 1996). "Direct measurement of the extravasation of polyethyleneglycol-coated liposomes into solid tumor tissue by in vivo fluorescence microscopy". International Journal of Pharmaceutics. 144 (1): 11–17. doi:10.1016/S0378-5173(96)04674-1.
- ^ Lilavivat S, Sardar D, Jana S, Thomas GC, Woycechowsky KJ (August 2012). "In vivo encapsulation of nucleic acids using an engineered nonviral protein capsid". Journal of the American Chemical Society. 134 (32): 13152–13155. doi:10.1021/ja302743g. PMID 22827162.
- ^ Ramishetti S, Huang L (December 2012). "Intelligent design of multifunctional lipid-coated nanoparticle platforms for cancer therapy". Therapeutic Delivery. 3 (12): 1429–1445. doi:10.4155/tde.12.127. PMC 3584330. PMID 23323560.
- ^ Jain RK (June 1987). "Transport of molecules in the tumor interstitium: a review". Cancer Research. 47 (12): 3039–3051. PMID 3555767.
- ^ Garcia J, Hurwitz HI, Sandler AB, Miles D, Coleman RL, Deurloo R, et al. (June 2020). "Bevacizumab (Avastin®) in cancer treatment: A review of 15 years of clinical experience and future outlook". Cancer Treatment Reviews. 86: 102017. doi:10.1016/j.ctrv.2020.102017. ISSN 0305-7372. PMID 32335505.
- ^ Li C, Jiang P, Wei S, Xu X, Wang J (July 2020). "Regulatory T cells in tumor microenvironment: new mechanisms, potential therapeutic strategies and future prospects". Molecular Cancer. 19 (1): 116. doi:10.1186/s12943-020-01234-1. PMC 7367382. PMID 32680511.
- ^ "Protein Kinase Inhibitors", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases, 2012, PMID 31643906, retrieved 2024-02-20
- ^ Tan HY, Wang N, Lam W, Guo W, Feng Y, Cheng YC (2018-02-19). "Targeting tumour microenvironment by tyrosine kinase inhibitor". Molecular Cancer. 17 (1): 43. doi:10.1186/s12943-018-0800-6. ISSN 1476-4598. PMC 5817793. PMID 29455663.
- ^ Mansour MA, Caputo VS, Aleem E (2021-11-01). "Highlights on selected growth factors and their receptors as promising anticancer drug targets". The International Journal of Biochemistry & Cell Biology. 140: 106087. doi:10.1016/j.biocel.2021.106087. ISSN 1357-2725. PMID 34563698. S2CID 237943611.
- ^ Kankeu Fonkoua LA, Sirpilla O, Sakemura R, Siegler EL, Kenderian SS (2022-06-16). "CAR T cell therapy and the tumor microenvironment: Current challenges and opportunities". Molecular Therapy - Oncolytics. 25: 69–77. doi:10.1016/j.omto.2022.03.009. ISSN 2372-7705. PMC 8980704. PMID 35434273.
- ^ Zhang C, Durer S, Thandra KC, Kasi A (2024), "Chimeric Antigen Receptor T-Cell Therapy", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 30725979, retrieved 2024-02-20
- ^ Brown CE, Aguilar B, Starr R, Yang X, Chang WC, Weng L, et al. (January 2018). "Optimization of IL13Rα2-Targeted Chimeric Antigen Receptor T Cells for Improved Anti-tumor Efficacy against Glioblastoma". Molecular Therapy. 26 (1): 31–44. doi:10.1016/j.ymthe.2017.10.002. PMC 5763077. PMID 29103912.
- ^ Schubert ML, Schmitt M, Wang L, Ramos CA, Jordan K, Müller-Tidow C, et al. (2021-01-01). "Side-effect management of chimeric antigen receptor (CAR) T-cell therapy". Annals of Oncology. 32 (1): 34–48. doi:10.1016/j.annonc.2020.10.478. ISSN 0923-7534. PMID 33098993.