BCR (gene)
 From Wikipedia the free encyclopedia
From Wikipedia the free encyclopedia
| Bcr-Abl oncoprotein oligomerisation domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
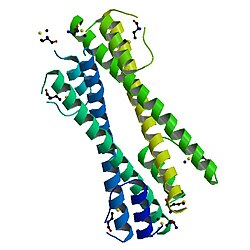
 structure of the bcr-abl oncoprotein oligomerization domain | |||||||||
| Identifiers | |||||||||
| Symbol | Bcr-Abl_Oligo | ||||||||
| Pfam | PF09036 | ||||||||
| InterPro | IPR015123 | ||||||||
| |||||||||
The breakpoint cluster region protein (BCR) also known as renal carcinoma antigen NY-REN-26 is a protein that in humans is encoded by the BCR gene. BCR is one of the two genes in the BCR-ABL fusion protein, which is associated with the Philadelphia chromosome. Two transcript variants encoding different isoforms have been found for this gene.
Function
[edit]Although the BCR-ABL fusion protein has been much studied, the function of the normal BCR gene product is still not clear. The protein has serine/threonine kinase activity and is a guanine nucleotide exchange factor for the Rho family of GTPases including RhoA.[5][6]
Clinical significance
[edit]A reciprocal translocation between chromosomes 22 and 9 produces the Philadelphia chromosome, which is often found in patients with chronic myelogenous leukemia. The chromosome 22 breakpoint for this translocation is located within the BCR gene. The translocation produces a fusion protein that is encoded by sequence from both BCR and ABL, the gene at the chromosome 9 breakpoint.[7]
Structure
[edit]
The BCR-ABL oncoprotein oligomerisation domain found at the N-terminus of BCR is essential for the oncogenicity of the BCR-ABL fusion protein. The BCR-ABL oncoprotein oligomerisation domain consists of a short N-terminal helix (alpha-1), a flexible loop and a long C-terminal helix (alpha-2). Together these form an N-shaped structure, with the loop allowing the two helices to assume a parallel orientation. The monomeric domains associate into a dimer through the formation of an antiparallel coiled coil between the alpha-2 helices and domain swapping of two alpha-1 helices, where one alpha-1 helix swings back and packs against the alpha-2 helix from the second monomer. Two dimers then associate into a tetramer.[8] Structure-based engineering starting from the antiparallel coiled coil domain of the BCR-ABL oncoprotein (BCR30-65) resulted in a new pH-sensitive homodimeric antiparallel coiled coil.[9]
Interactions
[edit]The BCR protein has been shown to interact with:
See also
[edit]References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000186716 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000009681 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Dubash AD, Koetsier JL, Amargo EV, Najor NA, Harmon RM, Green KJ (August 2013). "The GEF Bcr activates RhoA/MAL signaling to promote keratinocyte differentiation via desmoglein-1". The Journal of Cell Biology. 202 (4): 653–666. doi:10.1083/jcb.201304133. PMC 3747303. PMID 23940119.
- ^ "Entrez Gene: Breakpoint cluster region".
- ^ "Entrez Gene: BCR breakpoint cluster region".
- ^ Zhao X, Ghaffari S, Lodish H, Malashkevich VN, Kim PS (February 2002). "Structure of the Bcr-Abl oncoprotein oligomerization domain". Nature Structural Biology. 9 (2): 117–120. doi:10.1038/nsb747. PMID 11780146. S2CID 17453012.
- ^ Nagarkar RP, Fichman G, Schneider JP (2020-08-14). "Engineering and characterization of a<scp>pH</scp>-sensitive homodimeric antiparallel coiled coil". Peptide Science. 112 (5). doi:10.1002/pep2.24180. ISSN 2475-8817. S2CID 221920164.
- ^ a b c Puil L, Liu J, Gish G, Mbamalu G, Bowtell D, Pelicci PG, et al. (February 1994). "Bcr-Abl oncoproteins bind directly to activators of the Ras signalling pathway". The EMBO Journal. 13 (4): 764–773. doi:10.1002/j.1460-2075.1994.tb06319.x. PMC 394874. PMID 8112292.
- ^ Ling X, Ma G, Sun T, Liu J, Arlinghaus RB (January 2003). "Bcr and Abl interaction: oncogenic activation of c-Abl by sequestering Bcr". Cancer Research. 63 (2): 298–303. PMID 12543778.
- ^ Pendergast AM, Muller AJ, Havlik MH, Maru Y, Witte ON (July 1991). "BCR sequences essential for transformation by the BCR-ABL oncogene bind to the ABL SH2 regulatory domain in a non-phosphotyrosine-dependent manner". Cell. 66 (1): 161–171. doi:10.1016/0092-8674(91)90148-R. PMID 1712671. S2CID 9933891.
- ^ Hallek M, Danhauser-Riedl S, Herbst R, Warmuth M, Winkler A, Kolb HJ, et al. (July 1996). "Interaction of the receptor tyrosine kinase p145c-kit with the p210bcr/abl kinase in myeloid cells". British Journal of Haematology. 94 (1): 5–16. doi:10.1046/j.1365-2141.1996.6102053.x. PMID 8757502. S2CID 30033345.
- ^ a b c d Bai RY, Jahn T, Schrem S, Munzert G, Weidner KM, Wang JY, et al. (August 1998). "The SH2-containing adapter protein GRB10 interacts with BCR-ABL". Oncogene. 17 (8): 941–948. doi:10.1038/sj.onc.1202024. PMID 9747873. S2CID 20866214.
- ^ a b Million RP, Harakawa N, Roumiantsev S, Varticovski L, Van Etten RA (June 2004). "A direct binding site for Grb2 contributes to transformation and leukemogenesis by the Tel-Abl (ETV6-Abl) tyrosine kinase". Molecular and Cellular Biology. 24 (11): 4685–4695. doi:10.1128/MCB.24.11.4685-4695.2004. PMC 416425. PMID 15143164.
- ^ Heaney C, Kolibaba K, Bhat A, Oda T, Ohno S, Fanning S, et al. (January 1997). "Direct binding of CRKL to BCR-ABL is not required for BCR-ABL transformation". Blood. 89 (1): 297–306. doi:10.1182/blood.V89.1.297. PMID 8978305.
- ^ Kolibaba KS, Bhat A, Heaney C, Oda T, Druker BJ (March 1999). "CRKL binding to BCR-ABL and BCR-ABL transformation". Leukemia & Lymphoma. 33 (1–2): 119–126. doi:10.3109/10428199909093732. PMID 10194128.
- ^ Lionberger JM, Smithgall TE (February 2000). "The c-Fes protein-tyrosine kinase suppresses cytokine-independent outgrowth of myeloid leukemia cells induced by Bcr-Abl". Cancer Research. 60 (4): 1097–1103. PMID 10706130.
- ^ a b c Maru Y, Peters KL, Afar DE, Shibuya M, Witte ON, Smithgall TE (February 1995). "Tyrosine phosphorylation of BCR by FPS/FES protein-tyrosine kinases induces association of BCR with GRB-2/SOS". Molecular and Cellular Biology. 15 (2): 835–842. doi:10.1128/MCB.15.2.835. PMC 231961. PMID 7529874.
- ^ Million RP, Van Etten RA (July 2000). "The Grb2 binding site is required for the induction of chronic myeloid leukemia-like disease in mice by the Bcr/Abl tyrosine kinase". Blood. 96 (2): 664–670. doi:10.1182/blood.V96.2.664. PMID 10887132.
- ^ Ma G, Lu D, Wu Y, Liu J, Arlinghaus RB (May 1997). "Bcr phosphorylated on tyrosine 177 binds Grb2". Oncogene. 14 (19): 2367–2372. doi:10.1038/sj.onc.1201053. PMID 9178913. S2CID 9249479.
- ^ Stanglmaier M, Warmuth M, Kleinlein I, Reis S, Hallek M (February 2003). "The interaction of the Bcr-Abl tyrosine kinase with the Src kinase Hck is mediated by multiple binding domains". Leukemia. 17 (2): 283–289. doi:10.1038/sj.leu.2402778. PMID 12592324. S2CID 8695384.
- ^ Lionberger JM, Wilson MB, Smithgall TE (June 2000). "Transformation of myeloid leukemia cells to cytokine independence by Bcr-Abl is suppressed by kinase-defective Hck". The Journal of Biological Chemistry. 275 (24): 18581–18585. doi:10.1074/jbc.C000126200. PMID 10849448.
- ^ Radziwill G, Erdmann RA, Margelisch U, Moelling K (July 2003). "The Bcr kinase downregulates Ras signaling by phosphorylating AF-6 and binding to its PDZ domain". Molecular and Cellular Biology. 23 (13): 4663–4672. doi:10.1128/MCB.23.13.4663-4672.2003. PMC 164848. PMID 12808105.
- ^ a b Salgia R, Sattler M, Pisick E, Li JL, Griffin JD (February 1996). "p210BCR/ABL induces formation of complexes containing focal adhesion proteins and the protooncogene product p120c-Cbl". Experimental Hematology. 24 (2): 310–313. PMID 8641358.
- ^ Salgia R, Li JL, Lo SH, Brunkhorst B, Kansas GS, Sobhany ES, et al. (March 1995). "Molecular cloning of human paxillin, a focal adhesion protein phosphorylated by P210BCR/ABL". The Journal of Biological Chemistry. 270 (10): 5039–5047. doi:10.1074/jbc.270.10.5039. PMID 7534286.
- ^ Skorski T, Kanakaraj P, Nieborowska-Skorska M, Ratajczak MZ, Wen SC, Zon G, et al. (July 1995). "Phosphatidylinositol-3 kinase activity is regulated by BCR/ABL and is required for the growth of Philadelphia chromosome-positive cells". Blood. 86 (2): 726–736. doi:10.1182/blood.V86.2.726.bloodjournal862726. PMID 7606002.
- ^ Liedtke M, Pandey P, Kumar S, Kharbanda S, Kufe D (October 1998). "Regulation of Bcr-Abl-induced SAP kinase activity and transformation by the SHPTP1 protein tyrosine phosphatase". Oncogene. 17 (15): 1889–1892. doi:10.1038/sj.onc.1202117. PMID 9788431. S2CID 42228230.
- ^ Park AR, Oh D, Lim SH, Choi J, Moon J, Yu DY, et al. (October 2012). "Regulation of dendritic arborization by BCR Rac1 GTPase-activating protein, a substrate of PTPRT". Journal of Cell Science. 125 (Pt 19): 4518–4531. doi:10.1242/jcs.105502. PMID 22767509. S2CID 22422544.
- ^ Takeda N, Shibuya M, Maru Y (January 1999). "The BCR-ABL oncoprotein potentially interacts with the xeroderma pigmentosum group B protein". Proceedings of the National Academy of Sciences of the United States of America. 96 (1): 203–207. Bibcode:1999PNAS...96..203T. doi:10.1073/pnas.96.1.203. PMC 15117. PMID 9874796.
Further reading
[edit]External links
[edit]- BCR+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human BCR genome location and BCR gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: P11274 (Human Breakpoint cluster region protein) at the PDBe-KB.